When you add corn syrup to a mixture, it influences the overall freezing point of the solution, an effect known as freezing point depression. This is a principle you might encounter in the making of frozen desserts like ice cream or gelato.
The freezing point depression happens because the added solute—corn syrup in this case—disrupts the formation of ice crystals, which requires a lower temperature to initiate freezing. The specific impact of corn syrup on freezing point is tied to its unique properties compared to other sugars.

Corn syrup is composed largely of glucose and oligosaccharides, which give it distinct characteristics in how it affects the freezing point. One factor is its molecular weight; different types of corn syrup have varying average molecular weights, which correspond to how effectively they can lower the freezing point of a solution.
For example, a high-mean-molecular-weight corn syrup will affect the freezing point to a lesser degree than a corn syrup with a lower molecular weight. This is due to the number of particles in the solution: the more particles present, the greater the freezing point depression.
The mechanics of freezing point depression are not just a concern for those in culinary fields; they reflect fundamental physical chemistry principles. Grasping these concepts can illuminate why some frozen treats have a softer texture and why others are harder and more crystalline.
When you choose corn syrup as an ingredient in your recipes, you are leveraging its properties to manipulate the texture and consistency of your final product by affecting freezing point depression.
Fundamentals of Freezing Point Depression

Understanding the fundamentals of freezing point depression is essential for grasping how additives like corn syrup affect the freezing point of solutions. As you delve into this subject, you will uncover the relationship between solute concentration and the physical properties of a solution.
Definition and Principles
Freezing point depression occurs when a solute is dissolved in a solvent, causing the freezing point of the solution to be lower than that of the pure solvent. The freezing point is the temperature at which a liquid becomes a solid. In a solution, solute-solvent interactions hinder the formation of a solid structure, resulting in a decrease in freezing point. This behavior is a physical property that does not affect the chemical composition of the substances involved.
Freezing point depression is determined by the molality of the solution, which is the number of moles of solute per kilogram of solvent. Unlike molarity, molality is not affected by changes in temperature or pressure.
Furthermore, the nature of the solute plays a significant role. Nonelectrolyte solutions, which consist of molecules that do not dissociate into ions, have less impact on the freezing point compared to ionic compounds, or electrolytes, which dissociate into ions and contribute more to freezing point depression.
Colligative Properties and Their Effects
Colligative properties, such as freezing point depression, depend on the number of solute particles, rather than their identity. These properties include boiling point elevation, vapor pressure lowering, and osmotic pressure.
- Vapor Pressure: The addition of a non-volatile solute reduces the solvent’s vapor pressure at a given temperature.
- Boiling Point: As a consequence of lowered vapor pressure, the boiling point of a solution is higher than that of the pure solvent.
- Osmotic Pressure: A solution will “pull” solvent toward it through a semipermeable membrane, demonstrating osmotic pressure.
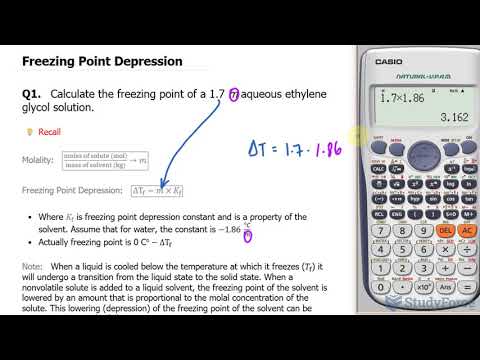
For each solute particle, the freezing point of a solvent will be lowered by a quantity known as the cryoscopic constant (Kf), specific to the solvent. The formula for calculating freezing point depression is:
ΔTf = Kf * m
- ΔTf is the change in freezing point.
- m is the molality of the solute.
It’s noteworthy that for solutions of electrolytes, you must consider the van’t Hoff factor (i), which accounts for the number of particles into which a compound dissociates. The revised formula considering ionic compounds becomes:
ΔTf = i * Kf * m
Corn Syrup Composition and Properties
Corn syrup plays a distinctive role in freezing point depression due to its unique composition of saccharides, which directly affects its physical characteristics and behavior in various solutions.
Constituent Saccharides
The primary saccharides in corn syrup are glucose and fructose, though it may also contain a minor amount of higher saccharides. Unlike sucrose, a disaccharide composed of glucose and fructose, the saccharides in corn syrup are primarily in their monosaccharide form. This affects how corn syrup interacts with water activity in food products.
- Glucose: A monosaccharide that is abundant in corn syrup.
- Fructose: Often found alongside glucose in varying ratios depending on the process used to create the corn syrup.
- Higher Saccharides: Present in smaller quantities and can include disaccharides like lactose.
Physical Characteristics of Corn Syrup
Corn syrup is characterized by its high viscosity and its hygroscopic nature, which enables it to retain moisture and control water activity effectively. It often has a lower average molecular weight compared to sucrose, which profoundly influences its colligative properties such as the ability to lower a solution’s freezing point.
- Viscosity: Corn syrup is notably viscous, which makes it particularly suitable for use in frozen desserts, helping to control texture.
- Hygroscopicity: Due to its composition, corn syrup is capable of binding water, thereby reducing the water activity in food products.
- Molecular Weights: The predominant monosaccharides in corn syrup have about half the molecular weight of sucrose, enhancing its freezing point depression capability when used in food products.
Corn Syrup in Aqueous Solutions
When you dissolve corn syrup in water, you are creating an aqueous solution that exhibits freezing point depression. This property significantly affects the solution’s behavior.
Corn Syrup as a Nonelectrolyte
Corn syrup is a nonelectrolyte, which means it does not dissociate into ions when dissolved in a solvent such as water. Your understanding of nonelectrolytes is crucial because they do not contribute to the conductivity of the solution but do affect properties like freezing points.
Due to its high molecular weight, corn syrup’s presence in an aqueous solution shifts the freezing point to a lower temperature compared to simple sugars.
Impact on Vapor Pressure and Freezing Point
The freezing point of a solution is depressed when a solute, like corn syrup, is added because it lowers the vapor pressure. The reduction in vapor pressure means that less energy (in the form of temperature) is needed for the solution to reach the phase transition from liquid to solid.
This is a colligative property, which is dependent on the concentration of the dissolved molecules. Higher concentrations of corn syrup will result in a greater depression of the freezing point.
Molecular Interactions and Solution Behavior
In an aqueous solution, the molecular interactions between the solvent (water) and the corn syrup result in a mixture with unique properties. The presence of glucose polymers within the corn syrup plays a key role, as these molecules interfere with the water’s ability to form a solid crystal lattice, hence lowering the freezing point.
The behavior of this solution can be predicted based on the molecular weight and the concentration of the corn syrup, affording you better control over processes like ice-cream production where texture and consistency are paramount.
Comparative Analysis of Solutes
In the study of freezing point depression, you’ll discover that various solutes, ranging from sugars to salts, influence the process differently. These differences are crucial for applications like food preservation where texture and stability are paramount.
Sugars Versus Salts
Sucrose, fructose, and glucose are common sugars that lower the freezing point of a solution by breaking into smaller particles. In comparison, NaCl (table salt) dissociates into two ions—sodium and chloride—when dissolved, generally causing a more significant freezing point depression compared to the sugars.
That’s because the number of particles in the solution—ions or molecules—directly correlates with the magnitude of freezing point depression, in accordance with Raoult’s law.
Here’s how you might typically see these solutes compared:
- Sucrose (C12H22O11): Disaccharide, does not dissociate in solution, less impact on freezing point.
- NaCl (sodium chloride): Salt, dissociates into two ions, more impact on freezing point.
- Fructose (C6H12O6): Monosaccharide, does not dissociate, less impact than salts.
- Glucose (C6H12O6): Monosaccharide, similar effect to fructose.
Corn Syrup Versus Other Sweeteners
Corn syrup is a syrup rich in saccharides, mainly composed of glucose. It affects the freezing point depression more subtly than simple sugars like sucrose due to its varying molecular weights—higher molecular weight means less effect on freezing point depression.
Sweeteners and their impact:
- Corn Syrup: High molecular weight, moderate freezing point depression.
- Alcohol: Lowers freezing point, but depends on the type of alcohol and its concentration.
- Sweeteners like sucrose: Typically cause less freezing point depression than corn syrup with equivalent concentration because they don’t break down into smaller molecules like corn syrup does.
Practical Implications of Freezing Point Depression

Understanding how substances like corn syrup affect freezing point depression is critical for various applications, particularly in the food industry and the creation of antifreeze solutions. Here, you will explore how this phenomenon is leveraged for optimization and product enhancement.
Food Industry Applications
In food formulation, you’ll find that corn syrup serves as an effective sweetener and stabilizer.
It plays a pivotal role in controlling ice recrystallization, which is essential in prolonging the storage time of frozen products.
The high concentration of glucose within corn syrup lowers the freezing point of the product by increasing the solute concentration.
This means that ice cream, for instance, remains softer at lower temperatures, enhancing its texture and scoopability.
- Osmotic Pressure: Corn syrup elevates osmotic pressure, reducing the amount of freezable water and helping in the preservation of texture.
- Solute Concentration: By increasing solute concentration, the risk of large ice crystal formation is minimized, providing a smooth mouthfeel in frozen desserts.
Antifreeze Solutions
Antifreeze solutions often contain ethylene glycol or glycerol, substances known for their freezing point depression capabilities.
When you are formulating an antifreeze, it’s vital to calculate the correct concentration of these compounds to ensure the solution remains liquid in cold environments.
This effect can be paralleled with the use of high-glucose corn syrup in food, as both contribute to lowering the freezing point through similar chemical principles.
- Ethylene Glycol: Typically used in automotive antifreezes, it effectively lowers the freezing point to protect engines in winter months.
- Glycerol: Found in non-toxic antifreeze formulations, glycerol is used for systems where toxicity is a concern, such as in food processing.
Theoretical Considerations
In this section, you’ll gain an understanding of the key theoretical principles that dictate how corn syrup influences the freezing point of a solution.
Knowing these fundamentals is necessary for practical applications and scientific analysis.
Freezing Point Depression Constant
The freezing point depression constant (K_f) is a property of the solvent that gives you the freezing point lowering per molal concentration of a non-volatile solute.
For any particular solvent, K_f is a set value. For water, the freezing point depression constant typically used is 1.86°C kg/mol.
When you dissolve solute molecules like corn syrup in water, the resulting mixture has a lower freezing point than pure water.
To gain a clearer sense of this phenomenon, consider the freezing point depression as a straight line on a graph, where the slope of this line corresponds to the K_f.
When dealing with dilute solutions, this relationship is mostly linear, meaning the change in freezing point is directly proportional to the number of moles of solute present.
Calculating Molal Freezing Point Depression
To calculate the freezing point depression (ΔT_f) caused by a solute such as corn syrup, you can use the formula:
ΔT_f = K_f * m
Where m is the molality of the solution, usually expressed in moles of solute per kilogram of solvent.
Since the molar mass of the solute affects the number of moles, calculating the accurate molality is crucial for your computations.
As an example, if you know the amount of corn syrup (with a specific molar mass) added to a solvent and the resulting freezing point, you can determine the depression of the freezing point.
Say you dissolve corn syrup with a known molar mass in water to create a 1 molal solution, then the freezing point will be depressed by 1.86°C.
In the context of corn syrup, which often contains a high concentration of glucose, a solute known for its freezing point depression properties, these calculations become particularly relevant as the syrup’s complex mixture of glucose molecules contributes to the overall effect.
Impact on Biological Systems
Corn syrup can significantly affect biological systems, particularly through interactions with cell membranes and osmotic forces, which are vital to organism survival.
Cell Membrane Interaction
Corn syrup, a nonelectrolyte with a high glucose content, permeates biological fluids and influences the cell membrane.
Its presence alters the salt concentration surrounding cells, which can lead to changes in osmotic pressure and water activity.
Your cell membranes depend on a delicate balance of these factors to function properly, maintain cell shape, and regulate the transport of substances in and out of the cell.
Osmotic Forces and Organism Survival
As corn syrup enters a biological system, it impacts osmotic forces by disrupting the balance of solutes.
The osmotic pressure generated due to a difference in salt concentration between the inside of the cell and the surrounding environment affects cell health and, consequently, organism survival.
Strong electrolytes, absent in corn syrup, dissociate completely and have a pronounced effect on osmotic balance, whereas nonelectrolytes like corn syrup can create diverse responses based on their concentration and molecular makeup.
Factors Affecting Freezing Point Depression
When considering how corn syrup influences freezing point depression, two critical aspects are the interactions between solute and solvent and the relationship of pH to the freezing point.
Solute and Solvent Variability
The type and concentration of solute you introduce into a solvent significantly impacts the degree of freezing point depression.
For instance, corn syrup, due to its high concentration of glucose, has a different influence on freezing point depression compared to sucrose.
Corn syrup can lower the freezing point more effectively because it contains multiple types of sugar molecules, primarily glucose, with a lower molecular weight than sucrose.
- Solvent: The chemical potential of the solvent is affected by the addition of a solute. In the case of corn syrup in water, water acts as your solvent, and its freezing point decreases as the concentration of corn syrup increases.
- Solute-Solvent Interactions: The interactions between corn syrup and water result in a physical phenomenon where the addition of corn syrup disrupts the hydrogen bonding in water, effectuating a lower freezing point.
It’s important to note that the degree of dissociation of the solute in the solvent also influences the freezing point. When solute molecules dissociate, they increase the particle count in solution, leading to greater freezing point depression.
pH and Freezing Point Relationship
The pH level of a solution can influence the freezing point to some extent.
Although not as prominent as solute concentration or solute-solvent interactions, variations in pH alter the chemical structure and interactions of the molecules present in a solution.
- Acidic or Basic Solutions: A change in pH could affect the solute’s state, solubility, and dissociation in the solvent.
- Chemical Reactivity: The reactivity of the solute due to pH changes could potentially affect the solute-solvent interactions, further manipulating the freezing and boiling points.
Experimental Insights

In the realm of food science, understanding how additives like corn syrup affect the freezing point of solutions is paramount.
You will discover how laboratory experiments can quantify the depression effect and what the data implies for food processing.
Laboratory Techniques
You begin by dissolving corn syrup in water to create solutions of varying molality—one measure of solute concentration, defined as moles of solute per kilogram of solvent.
Monitoring the change in freezing point as molality increases allows you to examine the colligative properties of the solution.
A typical laboratory setup might include a calibrated freezing point osmometer to accurately determine the point at which the solution transitions from liquid to solid.
The technique of recrystallization, where solutes are dissolved and crystallized under controlled conditions, is crucial in determining the purity and molar mass of the syrup’s constituents.
This is relevant since the molar mass affects the freezing point depression, according to the formula ΔT = iK_f(m), where i is the van’t Hoff factor, K_f is the freezing point depression constant, and m is the molality.
Data Analysis and Interpretation
Once you obtain your measurements, scrutinizing the data involves plotting the freezing point against molality.
By doing this, you will typically see a linear relationship in dilute solutions, where the mole fraction of corn syrup is low.
Mole fraction is another way to represent concentration, indicating the ratio of moles of corn syrup to the total moles in the solution.
As you interpret your data, remember that the presence of high-concentration glucose in corn syrup may influence the slope of the line since glucose has a significant effect on freezing point depression.
Your analysis would not only include looking for linearity but also considering deviations that could suggest complex interactions at a molecular level.
Such insights are consequential when deciding the appropriate concentration of corn syrup in frozen desserts, for example, to control texture and sweetness while preventing over-freezing.
Frequently Asked Questions
Corn syrup is known for its freezing point depression properties, which are crucial in the context of frozen desserts like ice cream.
Understanding its impact helps you optimize texture, control crystallization, and influence the shelf stability of your products.
How does the addition of corn syrup affect the freezing point of ice cream?
When you add corn syrup to ice cream, it lowers the mixture’s freezing point.
This occurs because the glucose and other sugars in corn syrup interfere with the formation of ice crystals, resulting in a softer texture even at lower temperatures.
What role does corn syrup play in the texture and consistency of frozen desserts?
Corn syrup contributes to a smoother and creamier texture in frozen desserts by hindering large ice crystals’ formation. This makes your frozen treats less icy and more enjoyable in texture.
Can corn syrup be used to control the crystallization of ice in frozen goods?
Yes, corn syrup can be effectively used to control ice crystallization in frozen goods.
Its composition, which includes higher glucose content, helps in managing and minimizing crystal size, ensuring a consistent texture throughout the dessert.
What are the comparative effects of using corn syrup versus other sweeteners on the freezing point depression in frozen treats?
Compared to other sweeteners like sucrose, corn syrup has a more significant effect on lowering the freezing point.
This is due to its lower molecular weight and higher fructose content, which makes it more effective in reducing crystal formation.
How does the sugar content in corn syrup interact with other ingredients to influence the overall freezing point?
The sugars in corn syrup interact with other ingredients by affecting the solute concentration in the mixture.
This alters the colligative properties, such as the freezing point, resulting in a decrease of the temperature at which the mixture begins to solidify.
To what extent does corn syrup impact the shelf stability and freezing qualities of commercial ice cream products?
Corn syrup has a positive impact on the shelf stability and freezing qualities of commercial ice cream products. It not only helps in maintaining a desirable texture over time but also prevents the formation of ice crystals.
Ice crystals are often associated with freezer burn.